Hemophilia Market Forecast to Reach USD 21.24 Billion by 2032 Driven by Gene Therapy Advances
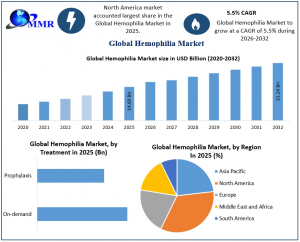
Hemophilia Market was valued at USD 14.60 Billion in 2025 & is projected to reach nearly USD 21.24 Billion by 2032, growing at a CAGR of 5.5% from 2026 to 2032.
The Hemophilia market is witnessing a historic pivot; Maximize Market Research identifies the transition from chronic management to one-time genetic cures as the industry's ultimate growth engine.”
ROCKVILLE , MD, UNITED STATES, March 5, 2026 /EINPresswire.com/ -- The global Hemophilia Market is entering a transformative era, shifting from chronic maintenance to long-term curative solutions. Valued at USD 14.60 Billion in 2025, the sector is projected to expand at a 5.5% CAGR, reaching USD 21.24 Billion by 2032. This growth is fueled by a structural transition toward gene therapies and non-factor replacement products that offer superior prophylactic protection. As healthcare systems prioritize value-based care, the demand for "once-and-done" genetic interventions is reshaping the competitive landscape. Maximize Market Research identifies this shift as the primary catalyst for market expansion, driving unprecedented R&D investment and redefining the standard of care for hemophilia patients worldwide.— Maximize Market Research
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/67045/
Therapeutic Dynamics: From Maintenance to Genetic Restoration
The standard of care is shifting from traditional Factor VIII/IX replacement toward next-generation non-factor therapies and AAV-vector gene transfers. While replacement therapy still commands the majority of the market, bispecific monoclonal antibodies like Hemlibra have revolutionized prophylaxis by reducing treatment burdens for patients with inhibitors.
A critical real-world validation of current market volatility is the recently announced strategic exit of Roctavian. In late February 2026, BioMarin confirmed it would voluntarily withdraw the therapy from the U.S. market by May 2026 following unsuccessful divestiture efforts. This move underscores significant hurdles in reimbursement and long-term durability that continue to challenge the USD 21.24 Billion landscape. Conversely, success stories like Hemgenix for Hemophilia B—which maintains factor expression in 94% of patients four years post-infusion—demonstrate the immense potential of curative "one-time" models. This divergence is pushing the industry toward rebalancing agents like Fitusiran, which aim to restore hemostatic balance through innovative antithrombin targeting.
Economic Perspective: Navigating the Million-Dollar Math
The hemophilia market’s economic structure is undergoing a radical shift from recurring revenue models to high-value, one-time curative payments. Maximize Market Research observes that while gene therapies carry an upfront cost—often reaching $3.5 million per dose—they offer a profound lifetime cost offset. In a typical severe hemophilia scenario, lifelong prophylactic factor replacement can exceed $20 million per patient, making curative interventions a "net-positive" for long-term payer sustainability.
A critical real-world validation of this economic pivot is the implementation of "Outcomes-Based Agreements" (OBAs) by payers in 2026. For example, CSL Behring has partnered with major U.S. insurers to offer refund guarantees if factor expression levels drop below a specific threshold within five years. This "value-over-volume" approach is accelerating the adoption of high-cost therapies and is a primary driver behind the market’s climb toward USD 21.24 Billion, as it provides the financial security necessary for broad-scale clinical integration.
Regional Ecosystems: Contrasting Global Growth Engines
North America remains the primary revenue stronghold, commanding over 45% of the global market due to high diagnostic rates and the rapid commercialization of gene therapies. However, the most significant shift is occurring in the Asia-Pacific region, which is projected to grow at a leading 6.5% CAGR through 2032. This surge is exemplified by the April 2026 WFH World Congress in Kuala Lumpur, which is currently serving as a catalyst for harmonizing treatment protocols across Southeast Asia. In markets like India and China, the "diagnosis gap" is rapidly closing as governments integrate hemophilia screening into national health missions, such as the expanded APBDR data collection initiatives aimed at improving patient registries. By transitioning from episodic care to a decentralized, specialty-clinic model, these emerging economies are creating the necessary infrastructure to support advanced prophylactic treatments, ensuring the global market remains on track to reach its USD 21.24 Billion valuation.
Segmentation Analysis: Strategic Value Distribution
The global landscape is fundamentally defined by the dominance of Hemophilia A, which commands approximately 81% of market revenue due to its higher prevalence and the rapid adoption of recombinant factor VIII. While Hemophilia B and C represent smaller patient pools, they are the primary targets for high-value Gene Therapy innovations, which are currently the fastest-growing therapeutic segment.
A critical shift is occurring in treatment regimens, where Prophylaxis has overtaken On-demand care as the standard of care in developed economies. This transition is fueled by the move from traditional Replacement Therapy to Non-factor and genetic interventions. Furthermore, Immune Tolerance Induction (ITI) therapy remains a vital, high-cost segment for managing patients with inhibitors. By integrating these diverse modalities from episodic on-demand infusions to "once-and-done" curative gene transfers the market is successfully addressing the full spectrum of disease severity, ensuring sustained expansion across all clinical end-user segments through 2032.
By Type
Hemophilia A
Hemophilia B
Hemophilia C
Others
By Treatment
On-demand
Prophylaxis
By Therapy
Replacement therapy
ITI therapy
Gene therapy
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/67045/
Recent R&D Milestones: The "Normalization" Frontier
The R&D pipeline is evolving beyond prophylaxis toward therapies that restore near-normal hemostatic function. A definitive 2026 milestone is the progression of Roche and Chugai’s NXT007, a next-generation bispecific antibody. In February 2026, Phase I/II data presented at the EAHAD Congress confirmed that patients could switch directly from Hemlibra to NXT007 without a washout period, achieving factor activity levels within the normal range.
Simultaneously, the regulatory landscape was reshaped by the March 2025 FDA approval of Sanofi’s Qfitlia (fitusiran). As the first siRNA therapy for hemophilia, it utilizes an antithrombin-lowering mechanism to "rebalance" the coagulation cascade with as few as six annual injections. Furthermore, Spark Therapeutics advanced its SPK-8011 gene therapy into the KEYSTONE 1 Phase III trial in early 2026, targeting durability exceeding five years. These breakthroughs in RNA interference and viral-vector delivery are transforming hemophilia from a lifelong burden into a manageable, and potentially curable, condition.
Analyst Perspective: The Durability Confidence Gap
The 5.5% CAGR reflects a profound paradigm shift where healthcare systems are increasingly valuing curative durability over chronic maintenance. Maximize Market Research highlights the 'Durability Confidence Gap' as the primary hurdle for gene therapy integration, where clinicians seek long-term endogenous expression data to justify upfront million-dollar price tags. However, the adoption of innovative Outcomes-Based Agreements is bridging this gap, transforming hemophilia from a lifelong variable expense into a predictable, one-time investment. By prioritizing lifecycle economics and the Net Present Value of avoided bleeds, the market is successfully de-risking advanced biologics. This strategic recalibration ensures that the transition to genetic restoration remains the definitive driver of the USD 21.24 Billion valuation.
Hemophilia Market, Key Players
Bayer AG
Bio Marin Pharmaceutical, Inc.
CSL Behring
Kedrion
Novo Nordisk
Pfizer, Inc.
Roche (Chugai Pharmaceutical Co.)
Sanofi (Genzyme Corporation)
Takeda Pharmaceutical (Shire Plc)
Shire (Baxalta)
Spark Therapeutics, Inc.
Genzyme Corporation
Chugai Pharmaceutical Co. Ltd.
Novo Nordisk A/S.
Octapharma AG
Get access to the full description of the report @ https://www.maximizemarketresearch.com/market-report/global-hemophilia-market/67045/
FAQ’s
What drives the USD 21.24 Billion Hemophilia Market?
Ans: The transition from on-demand care to prophylactic gene therapies is the primary catalyst, offering a lifetime cost offset against chronic factor replacement.
How do non-factor therapies impact growth?
Ans: Subcutaneous agents and bispecific antibodies are revolutionizing care for inhibitor patients, capturing high-value segments by reducing treatment burdens and breakthrough bleeds.
Why is Asia-Pacific the fastest-growing region?
Ans: A 6.5% CAGR is driven by aggressive diagnostic mobilization in India and China, closing the "diagnosis gap" through modernized screening and specialty clinics.
What role do Outcomes-Based Agreements play?
Ans: Refund guarantees for gene therapies are de-risking high-cost biologics, ensuring financial sustainability and steady clinical adoption through 2032.
Related Reports
Cranial Perforator Market: https://www.maximizemarketresearch.com/market-report/cranial-perforator-market/279576/
Dravet Syndrome Market: https://www.maximizemarketresearch.com/market-report/dravet-syndrome-market/278604/
Pediatric Palliative Care Market: https://www.maximizemarketresearch.com/market-report/pediatric-palliative-care-market/266684/
Pediatric Radiology Market: https://www.maximizemarketresearch.com/market-report/global-pediatric-radiology-market/103743/
Top Reports:
The Feminine Hygiene Products Market size was valued at USD 43.04 Billion in 2025 and the total Feminine Hygiene Product revenue is expected to grow at a CAGR of 6.43% from 2026 to 2032, reaching nearly USD 66.57 Billion By 2032. https://www.maximizemarketresearch.com/market-report/feminine-hygiene-products-market/221943/
Global Electronic Health Records Market size was valued at USD 29.98 Bn in 2023 and is expected to reach USD 48.90 Bn by 2030, at a CAGR of 7.24%. https://www.maximizemarketresearch.com/market-report/global-electronic-health-records-market/17991/
The MRI Market size was valued at USD 7.07 Billion in 2025 and the total MRI revenue is expected to grow at a CAGR of 6% from 2026 to 2032, reaching nearly USD 10.64 Billion by 2032. https://www.maximizemarketresearch.com/market-report/global-mri-market/8399/
The Surety Market size was valued at USD 23462.34 Million in 2025 and the total Surety revenue is expected to grow at a CAGR of 5.06% from 2025 to 2032, reaching nearly USD 33146.14 Million by 2032. https://www.maximizemarketresearch.com/market-report/surety-market/185094/
The Second-hand Product Market size was valued at USD 594.45 Billion in 2025 and the total Second-hand Product revenue is expected to grow at a CAGR of 13.6% from 2025 to 2032, reaching nearly USD 1451.34 Billion by 2032. https://www.maximizemarketresearch.com/market-report/second-hand-product-market/191282/
About Maximize Market Research
Maximize Market Research is a leading global market research and business consulting firm. Our revenue-impact and growth-driven initiatives empower Fortune 500 companies to navigate complex healthcare shifts and secure high-value market dominance.
Domain Focus: Hemophilia & Rare Disease
Our research evaluates high-performance therapeutic ecosystems through gene therapy innovation and non-factor replacement integration. We analyze the lifecycle economics and technological adoption shaping the global hematology landscape, from curative genetic restoration to sustainable value-based care frameworks.
Lumawant Godage
MAXIMIZE MARKET RESEARCH PVT. LTD.
+ +91 96073 65656
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.